Using CSA: Simian TB Serology Service when TB Threatens Research
When the Tuberculin Skin Test Fails: One Institution’s Experience with the Identification of TB in Research Cynomolgus Macaques.
Jay A, Marko S, Luke K, Ramos‐Rivera E, Culbreth M, Moreau A,Lugo‐Roman L, Bentzel D. In: Association Of Primate Veterinarians 46th Annual Workshop, 2018.
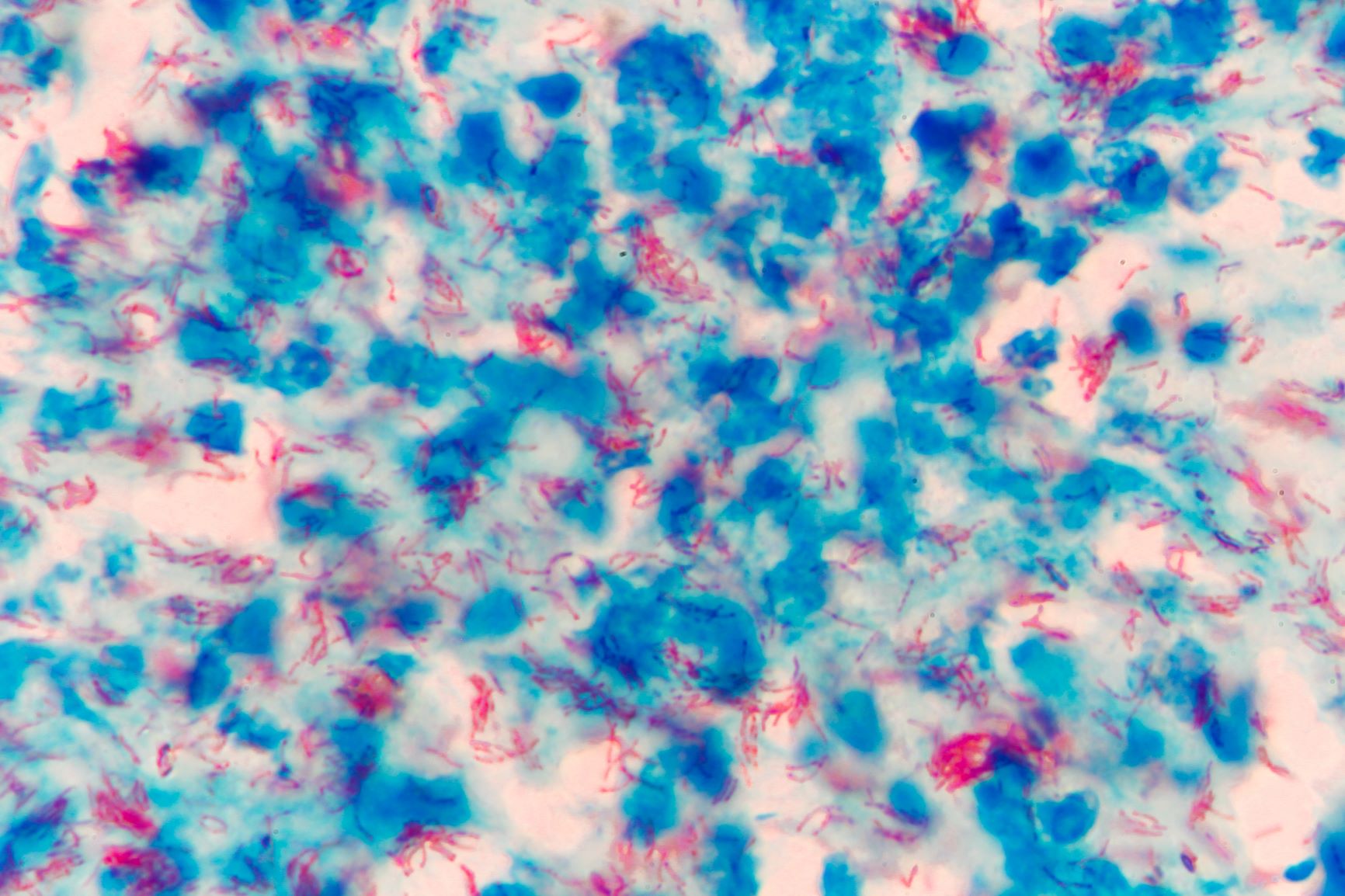
While mandatory screening by administration of intradermal tuberculin skin test (TST) in captive nonhuman primate colonies has resulted in significant reduction of Mycobacterium tuberculosis (M. tb) incidence, animals imported from countries with high rates of human infection still present a significant risk to research institutes and open colonies utilizing them. At a research institute in Maryland, U.S., cynomolgus macaques were identified with M. tb. Upon discovery of the first macaque with M. tb, room and building level quarantines, changes in personal protective equipment, consideration and employment of various diagnostic screening methods, retrospective postmortem lesion analysis, and enhanced employee tuberculosis (TB) screening were conducted. A test-and-cull strategy was established for cynomolgus macaques housed in the facility utilizing standard TSTs paired with serology conducted with a commercially available simian TB test. Antemortem serology yielded 6/120 animals positive for M. tb complex (M. tb, M. bovis, M. kansasii). All macaques that were euthanized had full necropsies, histopathological evaluation, PCR testing of tissue. Of the 6 that tested positive, 1 had significant gross lesions that were positive on histopathological evaluation, PCR and culture positive M. tb. Post-mortem serology using stored samples revealed an additional 1/10 animals positive with M. tb complex. A retrospective survey of histopathological evaluations of animals necropsied within the last 2 y revealed an additional 6 animals with granulomatous lesions and when tested for M. tb with immunofluorescent staining were positive. TST failed to identify any positive animals in the colony at any time. Improved Tb surveillance, beyond TST monitoring, should be considered for high risk groups of nonhuman primates. Incorporating serology as a component of a TB screening program for cynomolgus macaques may prevent the loss of animals, disruption of research, occupational health risk to personnel, and overall economic burden related to outbreak and disease control.